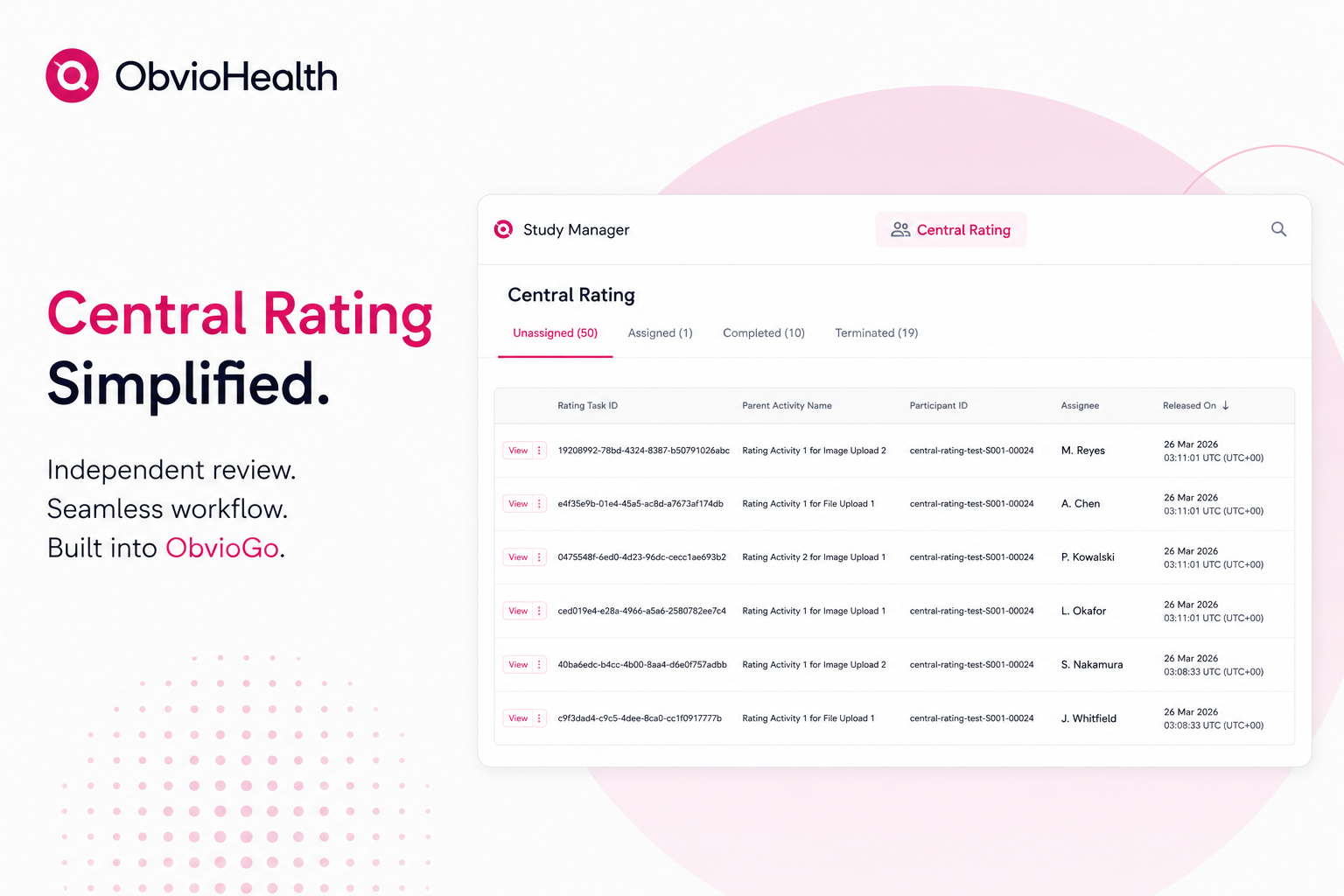
How eConsent Can Make the Difference in Clinical Trials
eConsent can mitigate these shortcomings by providing flexibility and engagement that empower patients with information and sponsors with data integrity.
Digital technologies have transformed the clinical trials industry. But, the informed consent process still remains predominantly paper based. Paired with the complexities of trial design and dynamic regulations, the inefficiencies of the traditional consent process pose a slew of problems—all of which can diminish data quality and study validity.
Electronic consent (eConsent) can mitigate these shortcomings by providing flexibility and engagement that empower patients with information and sponsors with data integrity.
Read on to understand the unique ways eConsent can be optimized for better trial outcomes.
Challenges of the Traditional Informed Consent Process
In an ideal setting, informed consent is the start of a relationship between the researcher and the participant. The study coordinators and the principal investigators (PIs) provide prospective participants with details about the trial and their responsibilities in it, followed by an interactive Q&A session. In a real-world setting, however, the study team is often under immense time pressures, and, as a result, detailed conversations to confirm participants’ comprehension is often deprioritized.
No matter the hurdles, informed consent documentation is still a foremost consideration in clinical study audits performed by the Food and Drug Administration (FDA). Failure to comply with regulatory and protocol requirements for informed consent can result in the “revocation of investigation privileges,” as occurred in the tragic 2001 trial conducted by John’s Hopkins University. Yet, despite the high stakes, research shows that inadequate ICFs are among the most frequently cited deficiencies resulting from FDA inspections.
To compound the regulatory risk, flaws in the traditional informed consent process can also have detrimental effects on a study’s validity. Participants who cannot demonstrate reasonable understanding of trial requirements run the risk of not adhering to the study’s protocol. Research also suggests that they are more likely to drop out of a trial, because they didn’t fully understand their roles from the beginning. Overall, a lack of participant understanding can compromise participant safety, as well as data accuracy, patient retention, engagement, and sample size.
Designing eConsent to Mitigate the Failures of the Traditional Process
By removing site visits for consent, the virtual format is definitively more convenient for participants. But, it’s not more engaging unless it’s designed to capture and keep participants’ attention.
eConsent should be designed to:
1. Ease the user's experience
- Key components and complex passages of the ICF can be animated or presented in digestible portions
- Self-guided videos can lead patients through key aspects of the consent form
2. Ensure participant comprehension
- Features can be implemented to control the speed at which participants scroll through consent documents
- Quizzes can be used throughout the consent and clinical trial process to assess participants’ comprehension
3. Provide better access to information
- Access to a digital repository of updated ICFs signed by the participants increases transparency
- Trial teams can ensure that participants have signed the amended document by making their signature mandatory before accessing further trial tasks
The Benefits of eConsent
The COVID-19 pandemic fast-tracked the adoption of decentralized clinical trials (DCTs), resulting in a seismic shift and accelerating the adoption of eConsent. The benefits of this span every touchpoint of clinical trials, optimizing the process for participants, sponsors, and regulatory bodies alike.
When designed to engage, eConsent empowers study participants with the knowledge they need to make optimal choices for themselves, and it transforms an often-overwhelming process into an engaging experience. By reducing or eliminating the need for on-site visits, eConsent is also more convenient for participants, allowing them to process the material at their own pace and consult friends and family.
On the sponsor side, research demonstrates interactive eConsent formats improve participant comprehension and satisfaction. This results in higher patient retention and compliance and ultimately better data.
Finally, eConsent supports the auditing and review processes of regulatory bodies, such as the FDA or internal review boards (IRBs). Digital ICF repositories increase transparency and, in turn, reduce inspection findings. And, by prioritizing participant understanding, eConsent improves the safety and protection of patients, which is concomitant with the priorities and key functions of regulatory bodies.
eConsent is Transforming the Clinical Trial Experience
eConsent isn't about simply digitizing the traditional process. Instead, this small but essential step that initiates the patient into a trial is fundamentally improving trial delivery. By reducing the burden on participants and ensuring they are better informed, eConsent makes them more likely to stay retained, engaged, and compliant —a win for all stakeholders.