Nuestros recursos
A continuación encontrará estudios de casos, seminarios web, blogs y comunicados de prensa en los que compartimos información valiosa sobre los ensayos clínicos descentralizados.
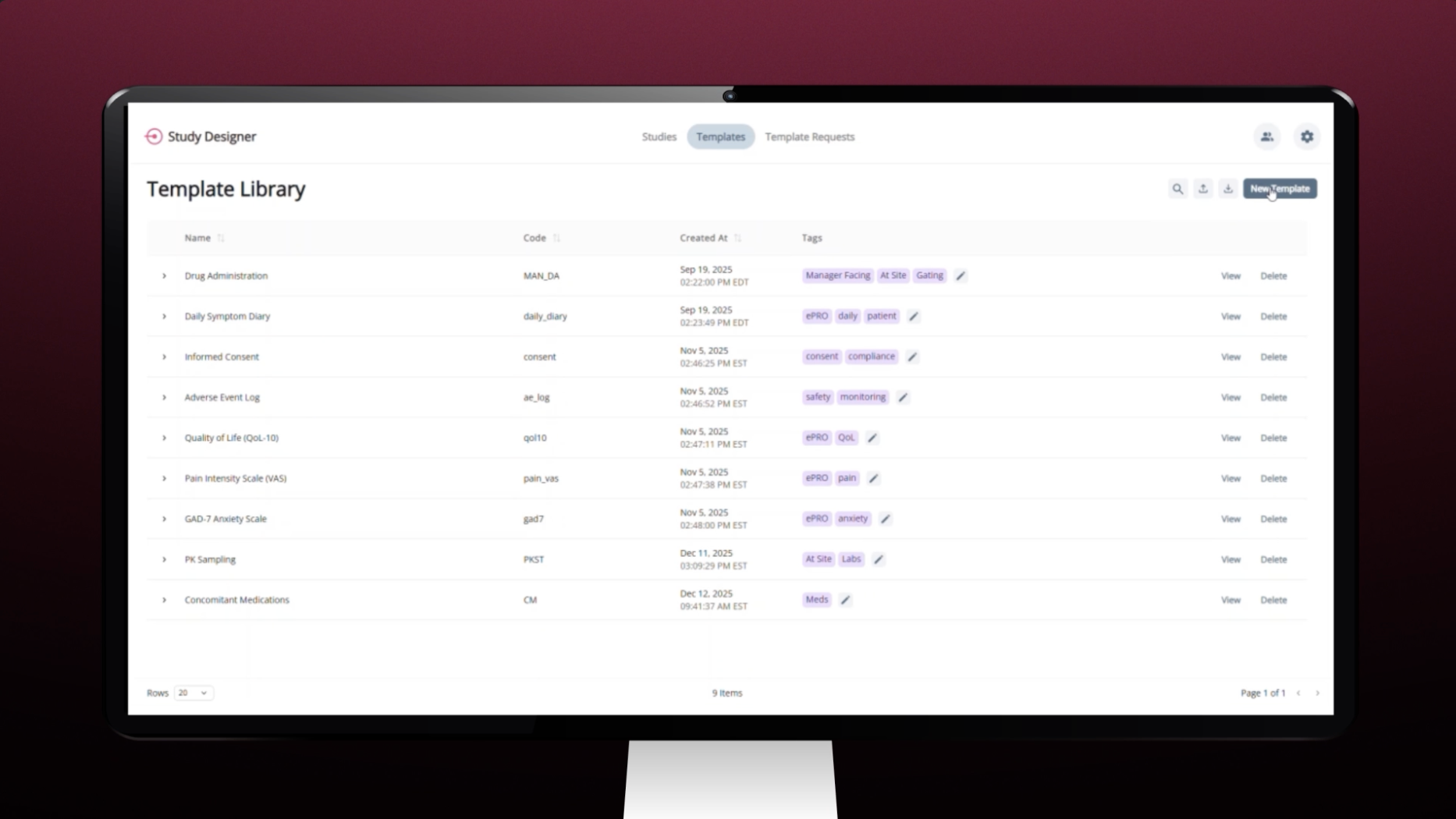
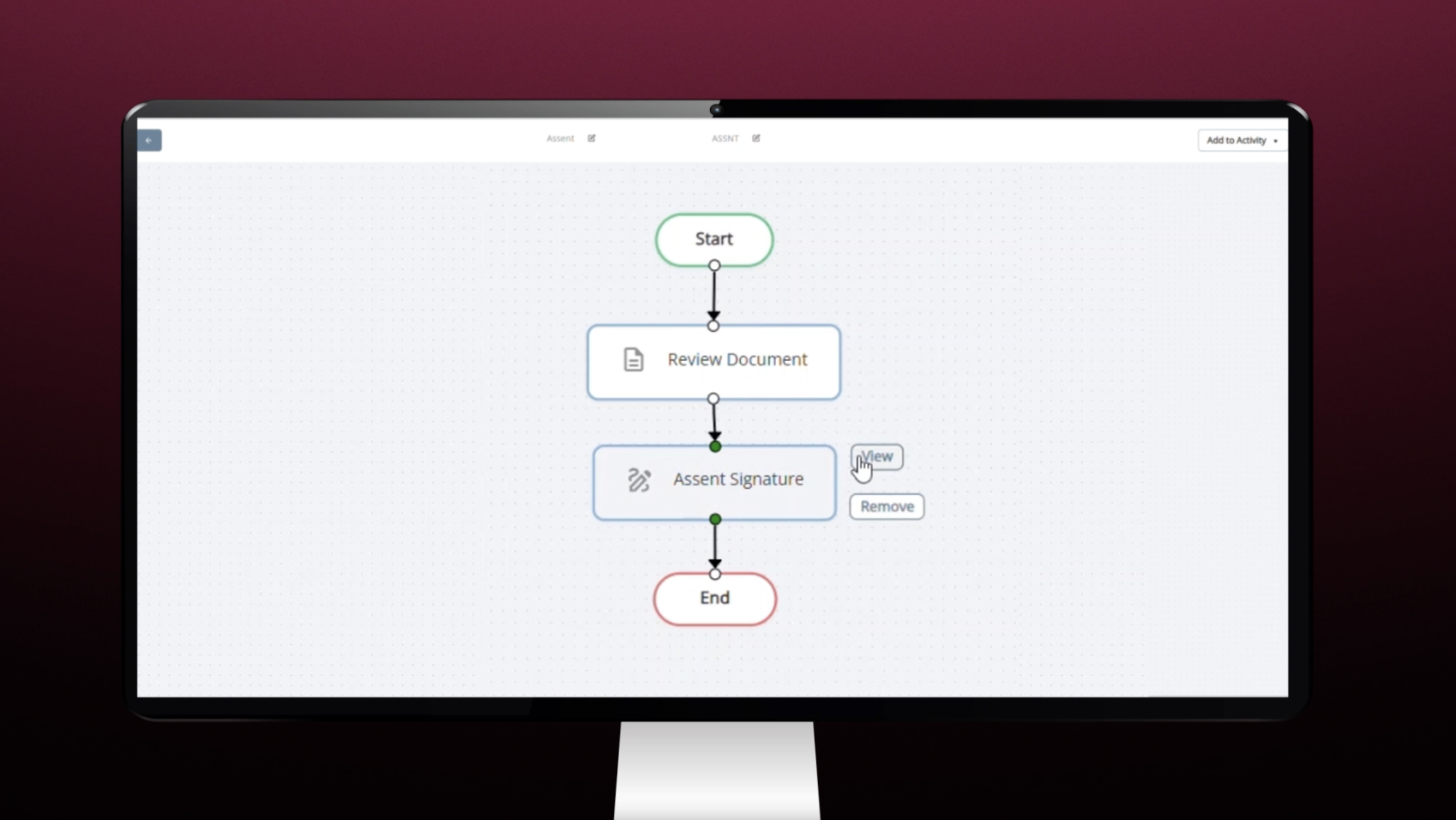
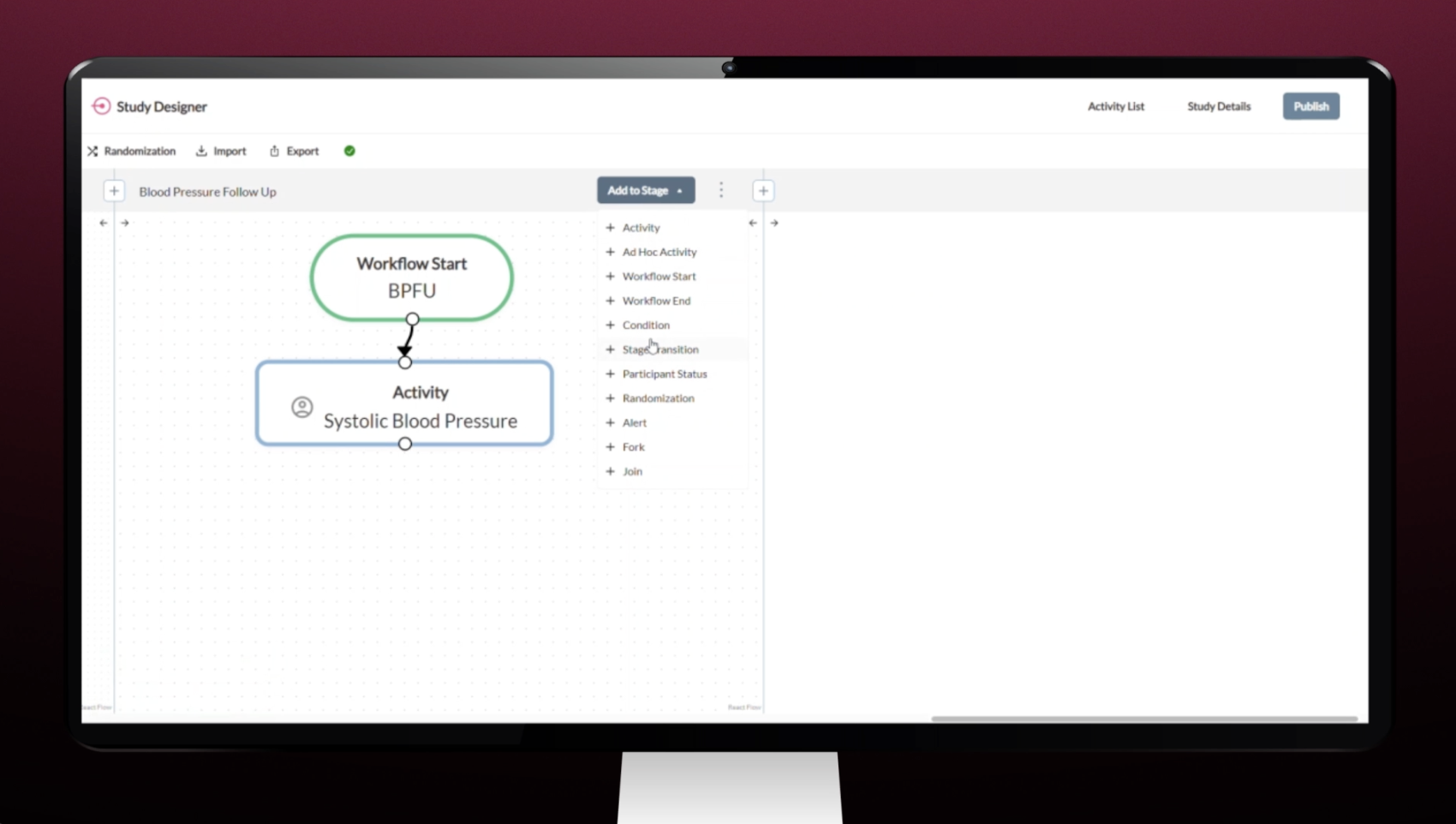
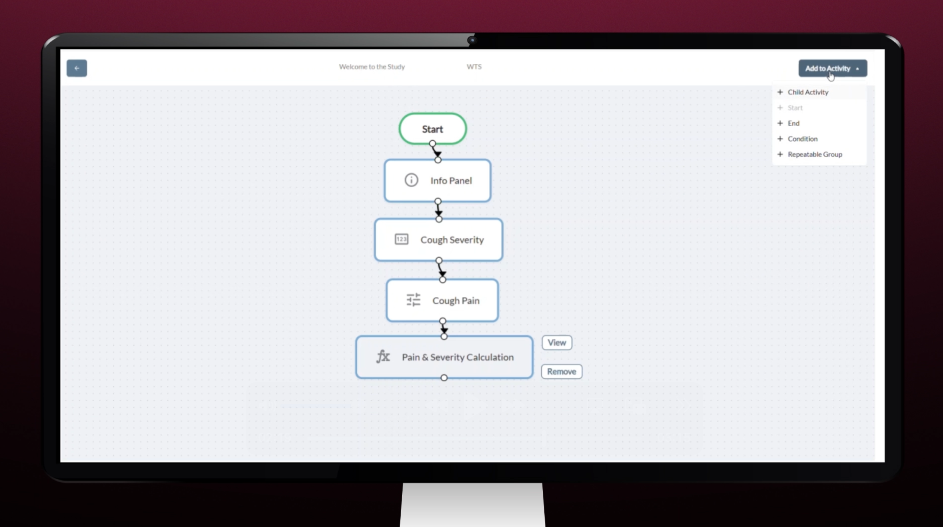
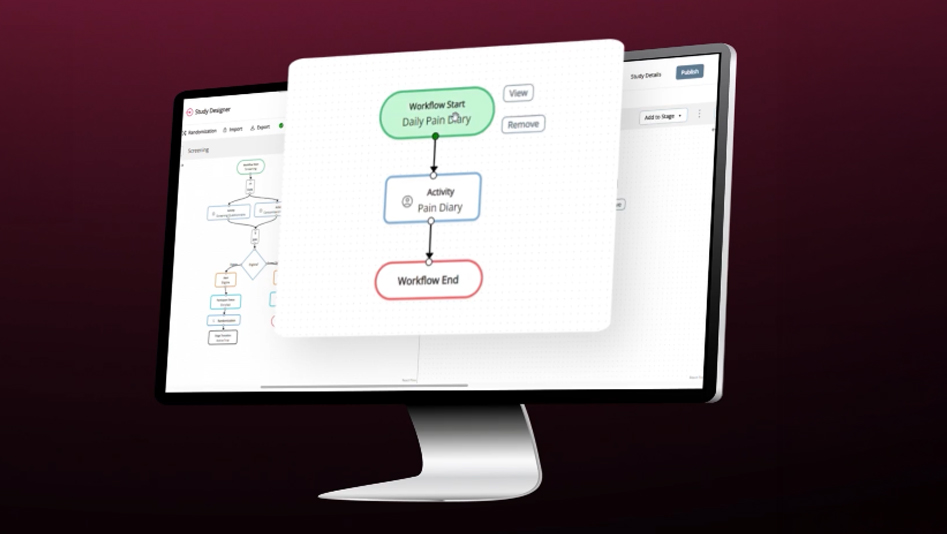
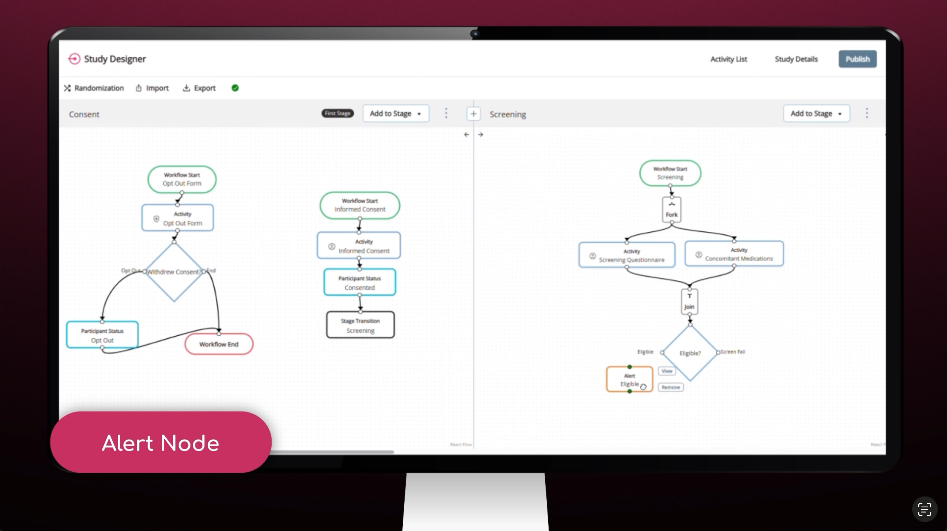
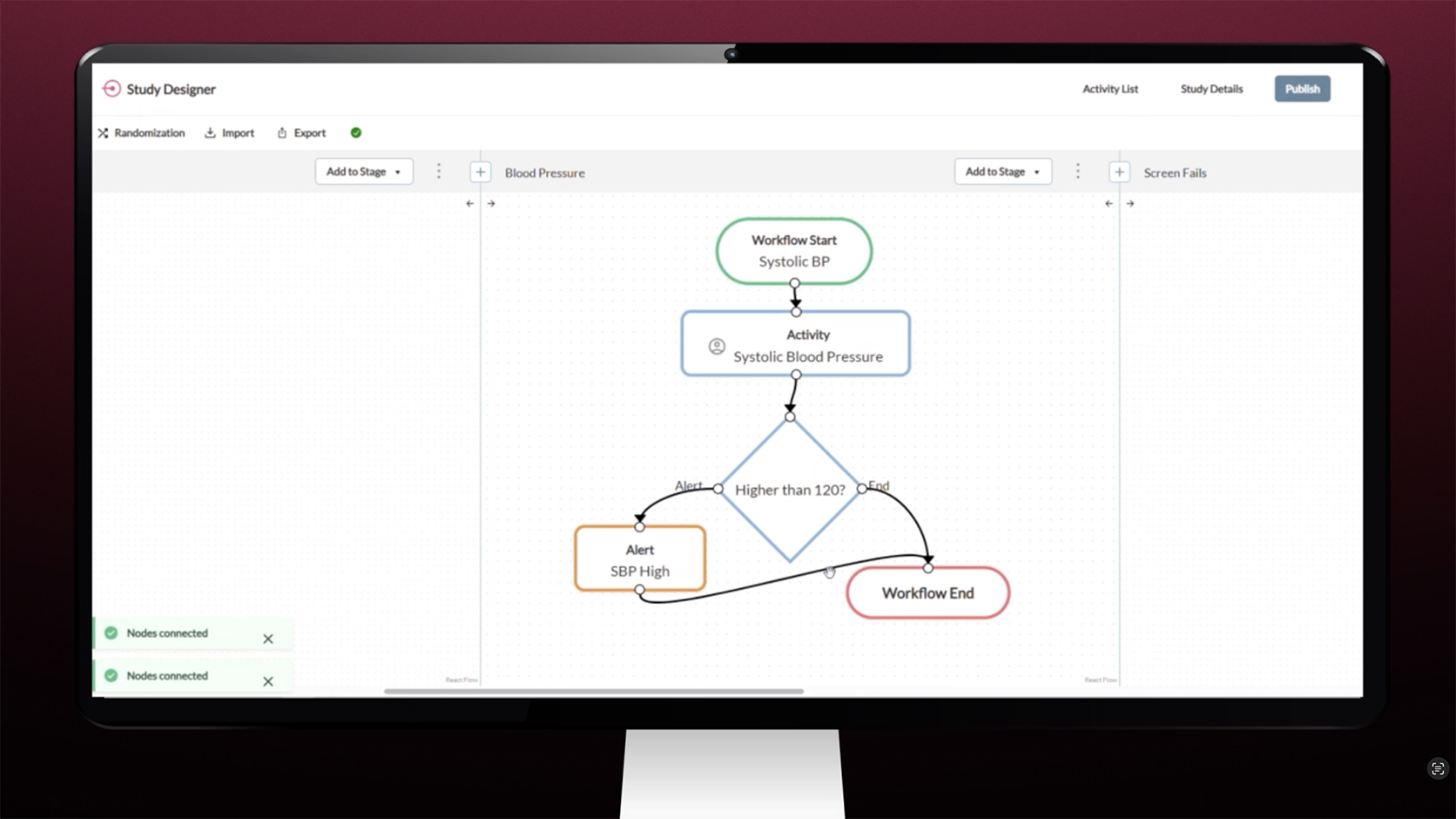
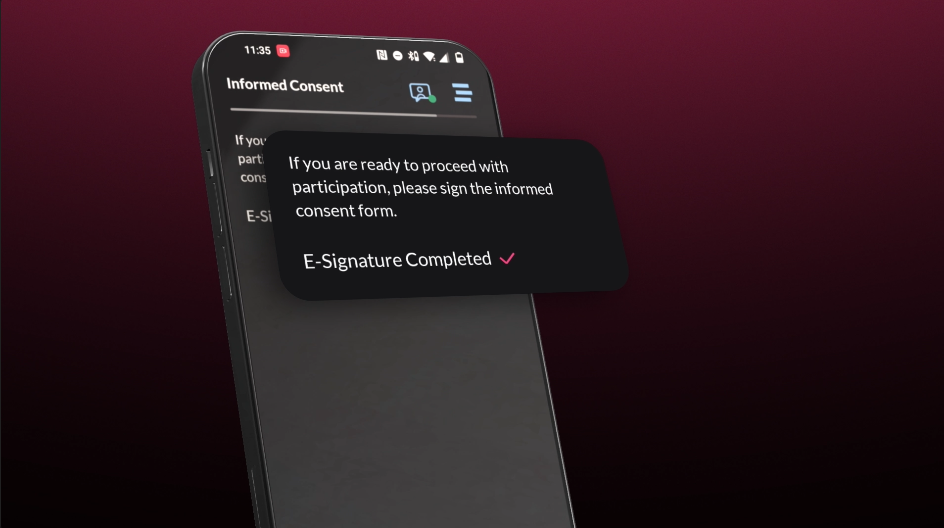
Study Designer
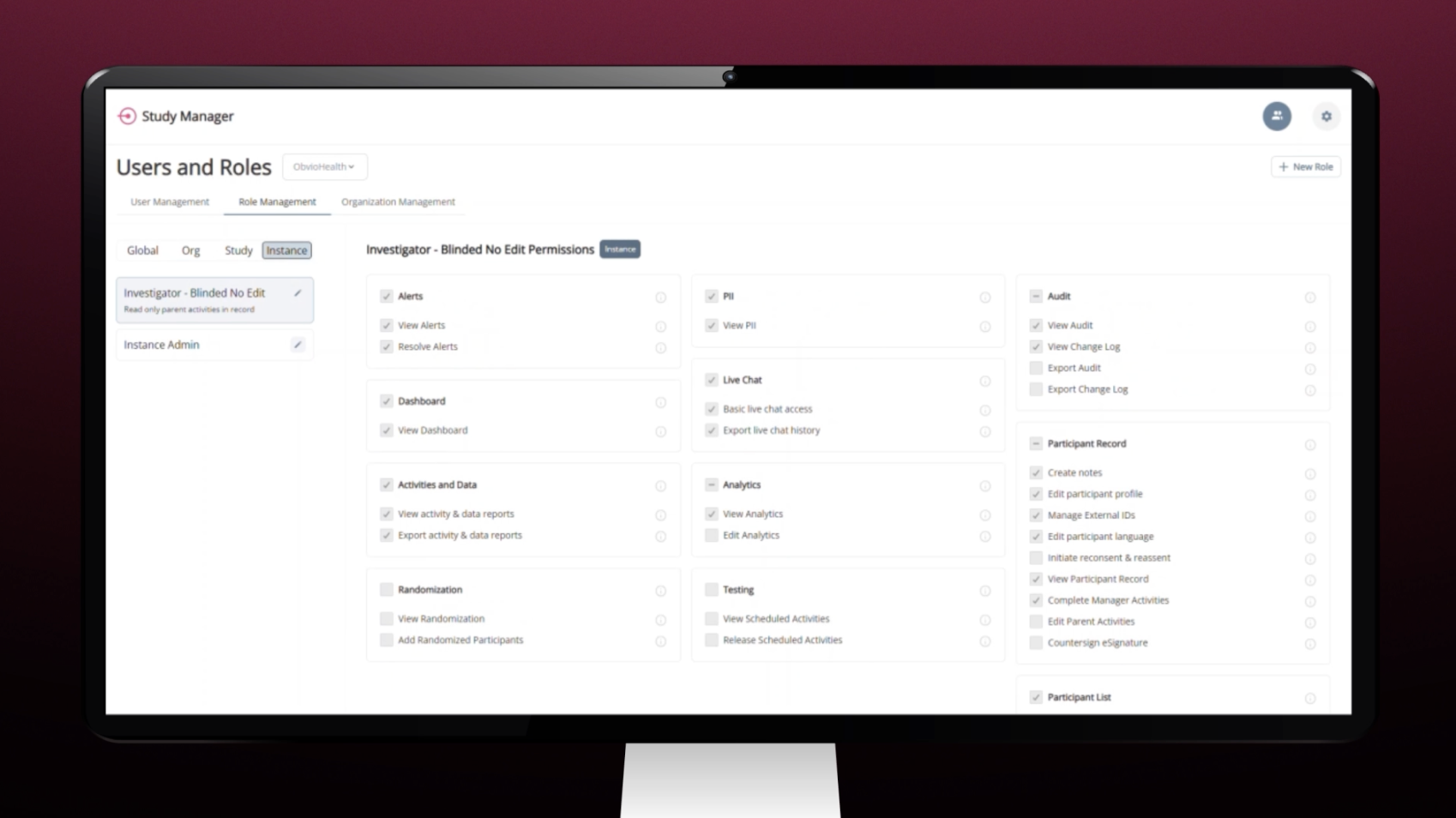
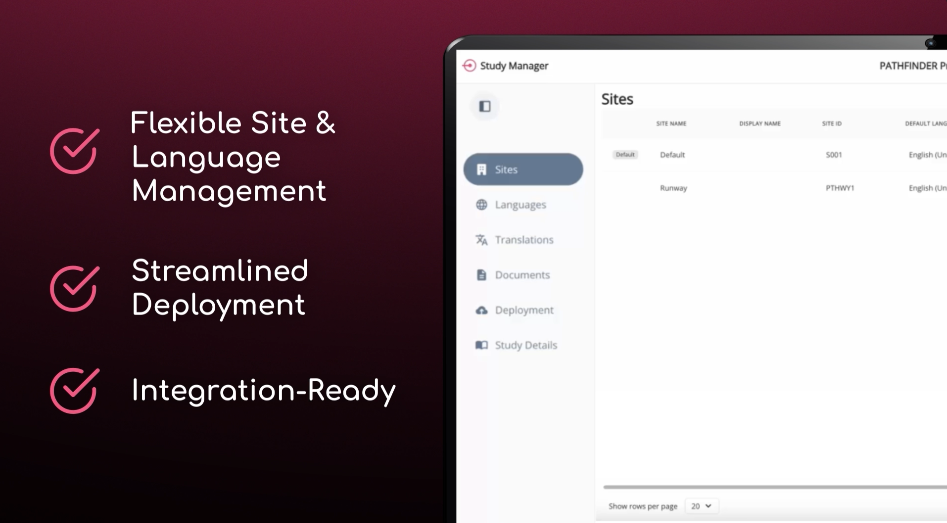
Study Manager
Thank you! Your submission has been received!
Oops! Something went wrong while submitting the form.