How Virtual Clinical Trials are Revolutionizing Health Research
Virtual clinical trials (VCTs) are a transformative approach to clinical research that's gaining momentum in the life sciences industry.
What Stakeholders Need to Know About VCTs
This article will explore the transformative possibilities of VCTs, from enhancing patient engagement to streamlining research processes and, ultimately, improving health outcomes. As we navigate this promising landscape, we will explore what stakeholders need to know to reap the benefits of VCTs in their research plans.
Understanding Virtual Clinical Trials
Virtual clinical trials, often referred to as decentralized trials, are a relatively new addition to the field of health research.1 They leverage digital tools and technologies to collect data from participants remotely, in their natural living or working environments. This contrasts with traditional clinical trials, where participants are required to visit research facilities or clinics at regular intervals for data collection and monitoring. Virtual trials can be conducted either fully remotely or using a hybrid approach.2 Fully remote trials are run entirely away from clinics, with all participant interactions happening either at, or in proximity to, their homes. In contrast, hybrid trials blend traditional in-person visits to sites with remote elements.
VCTs rely on a broad range of digital tools used to engage participants, manage data, and ensure that trials run smoothly. The key VCT elements are briefly summarized below.
eConsent
eConsent is a digital method of obtaining informed consent from participants in clinical trials. It uses interactive, multimedia tools to provide information and gather participants’ consent electronically. By allowing patients to review study protocols at their own pace, eConsent improves transparency, enhances patient understanding, and ensures regulatory compliance. It streamlines the administrative process, reducing the time and resources required versus paper-based consent. It also facilitates reconsenting when protocol changes arise.
ePRO
Electronic patient-reported outcomes (ePRO) are health outcomes or behaviors directly reported by the patient and collected electronically. ePRO tools capture data about a patient's activities and health status, including symptoms, functionality, and quality of life. In the past, ePROs were commonly captured through provisioned devices. VCTs provide for bring-your-own-device (BYOD) ePRO models, where the patients use their own smartphone devices to capture and submit the data. This offers real-time, accurate data collection, increasing the reliability of patient-reported data in trials.
Telemedicine
Telemedicine plays a significant role in VCTs. It enables remote patient consultation and assessments, reducing the need for in-person visits and making participation more convenient for patients. By offering flexibility and access, telemedicine can increase participant recruitment and retention rates in clinical trials.3
Sensors and Wearable Devices
Sensors and wearable devices offer new opportunities for data collection in VCTs. These devices can episodically or continuously monitor various health parameters—including heart rate, sleep patterns, activity levels, and more—providing rich, objective data in real time.4 This technology allows researchers to gain more comprehensive insight into a participant's health and response to treatment, ultimately enhancing the quality of trial data.5
Remote Patient Monitoring
Remote patient monitoring is key to virtual trials because it ensures the timely oversight of possible safety issues (adverse or serious adverse events) so proper actions can be taken quickly. Study teams can receive regular alerts to compliance lapses, as well as prompts to follow up with patients to encourage them to reengage. The real-time nature of remote monitoring can capture and address issues more quickly than when communication is reserved only for site visits.
The use of eConsent, ePRO, telemedicine, wearable devices, and remote patient monitoring enables patients to participate in trials from their natural environments, allowing for more convenient, accurate, and efficient data collection.
As VCTs continue to evolve, we will see more ways to leverage technology to offer a more inclusive, efficient, and patient-centric approach. But, what does this mean for the various stakeholders involved in clinical trials? What promise do these novel trial methods hold for patients, sponsors, and clinical sites?

The Promise of Virtual Clinical Trials
As we delve into the significant transformation VCTs offer in the field of health research, it's essential to highlight the main benefits across different user populations. The convenience and accessibility advantages for patients, the improved recruitment and cost-efficiency for sponsors, and streamlined processes for clinical sites are just a few examples. Indeed, the advent of VCTs represents a paradigm shift in clinical research, breaking down the main barriers to participation: geography and logistics. The ensuing discussion offers greater insight into the impacts on each stakeholder group.
The Patient Perspective: Accessibility and Convenience
One of the key advantages of VCTs lies in their accessibility. The elimination of geographical constraints opens a world of opportunities for patients—irrespective of their location, ethnicity, or race. This democratizes clinical trials, increasing the likelihood that they will be representative of the populations they are intended to serve.6 In some cases, this may mean greater ethnic or racial diversity. In others, it may enable more rural representation. And, in others, it may allow groups with less mobility to participate. Additionally, virtual clinical trials are particularly key to enabling the recruitment of populations for rare disease studies, where a broader geographic scope is essential to obtaining sufficient sample size.6(p318)
VCTs also improve the patient experience by reducing the logistical burdens associated with traditional trials. From enabling virtual enrollment to data capture and contact with study teams, every step of the process is made simpler and more convenient.
Patient benefits are aptly encapsulated in the words of FDA Commissioner Robert M. Califf, MD, who stated that VCTs can “enhance convenience for trial participants, reduce the burden on caregivers, expand access to more diverse populations, improve trial efficiencies, and facilitate research on rare diseases and diseases affecting populations with limited mobility.”7
Sponsor Benefits: Improved Recruitment, Accuracy, and Cost-Efficiency
From a sponsor’s perspective, VCTs are already proving to be a game-changer. For one, they have significantly improved recruitment and enrollment rates.8 By reaching a wider audience, VCTs make it easier to bring more people into studies more easily versus restricting recruitment to only a few locations in proximity to research sites. It is currently estimated that only 5% of adults in the U.S. have participated in a trial.9 VCTs should enable this number to climb significantly. By making trials easier and reducing travel requirements, VCTs can also enhance patient engagement, leading to higher trial compliance and completion.
Additionally, VCTs can provide more accurate evidence. Thanks to the real-time, real-world data gathering capabilities of digital technologies, the data collected is often less biased and more comprehensive, leading to better substantiation of efficacy and safety endpoints.10
On the cost front, the efficiencies achieved with VCTs can translate to significant financial savings. Reduction in travel expenses, site infrastructure needs, and investigator burden contribute to these savings, making trials more cost-effective.11
Clinical Site Advantages: Streamlined Processes and Monitoring
VCTs offer numerous benefits for clinical sites; primary among them is the reduction in administrative burden. By streamlining and automating various processes, VCTs free up valuable resources, allowing site administrators to focus on critical aspects of patient care.12 Digital tools used in VCTs also make safety and compliance monitoring easier, allowing for clearer insight into a patient's condition between visits.13 This ensures that any deviations from the expected outcomes can be detected and addressed promptly.
Recognizing and Addressing Virtual Clinical Trial Challenges
Though virtual clinical trials (VCTs) have emerged as a promising evolution in health research, this new approach to trials also comes with its own challenges. The issues most frequently cited include the digital divide, data privacy, the evolving regulatory environment, and appropriateness for complex or high-risk trials.14 This section reviews these challenges and offers possible solutions to enable the potential of VCTs to be fully harnessed.
Bridging the Digital Divide
One of the critical challenges of VCTs is the flip side of its greatest strength: These studies have a heavier reliance upon technology. Yet, not every patient has ready access to all technologies, thus highlighting the need to reduce the gap between the tech haves and the have-nots. This divide is notably more present among rural communities, minorities, and low-income groups.15
Possible Solutions
- Provision of Devices and Internet Services: Participants can be provided with the necessary devices and broadband internet services to help ensure that everyone, regardless of their economic status or location, can participate in VCTs.
- Community Partnerships: Partnerships with community centers or local libraries can facilitate access to the internet and devices necessary for participation in VCTs.
- Education and Training: Training can be implemented to ensure participants understand how to use digital technology effectively. This can empower individuals, particularly older adults who may not be as technologically adept.
- Access to Help Desks: It is important that participants have access to individuals who can help them when a technical issue arises, whether it be difficulty downloading an app, connecting a device, or following a usage instruction.
Data Privacy and Security Concerns
The reliance on digital tools and data infrastructures to support VCTs does generate data protection challenges.14 Sponsors and CROs must stay abreast of the global patchwork of data privacy requirements and cybersecurity risks, and have systems in place to ensure compliance and protection. Interestingly, sponsors often assume that concerns about data privacy are a key barrier to patient participation in virtual trials. However, a recent NIH review does not support this hypothesis.16 Nonetheless, there is a need for stringent privacy-preserving technologies to safeguard against unauthorized data uses.
Possible Solutions
- Pseudonymization and Data Minimization: Techniques like pseudonymization and data minimization can help ensure that personal data is not readily identifiable.
- Security Audits: Regular security audits can help identify and address vulnerabilities before they can be exploited.
- Patient Education: Educating patients on data privacy, and how their data will be used, is important in building trust while ensuring the protection of any sensitive data from unauthorized access.
An Evolving Regulatory Environment
The COVID-19 pandemic accelerated the need to evolve the regulatory frameworks surrounding virtual clinical trials. The FDA was the first to establish new guidelines.17 Other regions are still grappling with the best way to navigate this new approach to research. Sponsor concerns about a lack of clear and established guidelines can slow adoption.
Possible Solutions
- Focus on Best Practices: The guidance issued to date has been supportive of virtual clinical trials that follow best clinical practices. Sponsors should carefully consider the ways in which their studies can ensure patient safety, the rigorous collection of data, and the delivery of meaningful outcomes.
- Collaborate with Regulatory Bodies: Ethics committees and regulatory authorities are eager to collaborate with sponsors early in the clinical trial journey to discuss the best path forward for a given protocol. Active engagement with these bodies can help shape the development of guidelines that are practical and effective.
Ensuring Appropriate Trial Design
Not all clinical trials are suited to a virtual approach. Trials involving high-risk patients or complex interventions may require frequent in-person supervision and care.
Possible Solutions
- Careful Protocol Review: Every study is unique. Determining the right design requires deep understanding of the patient population, desired endpoints and outcomes, and trial risks. It is important to work with digitally savvy clinical trial experts who are able to design an approach that is most suitable for the population, condition, and treatment under study—leveraging digitization and/or decentralization as appropriate.
- Think Hybrid: When complex or high-risk trials require frequent in-clinic visits, there may still be certain trial components that can be virtualized. Contact with study coordinators and electronic reporting between visits through an app can complement traditional visits and deliver better outcomes. Here, too, engaging with experts experienced in virtual clinical research can be extremely beneficial.
Virtual clinical trials represent a significant advancement in clinical research. Increased patient accessibility, improved recruitment, cost-efficiency, and streamlined processes offer a compelling case for the adoption of these innovative methods. However, as with any groundbreaking approach, the promises of VCTs are balanced by an array of novel challenges that the healthcare industry needs to recognize and address. While the technology that enables these trials continues to improve, certain socioeconomic, regulatory, and practical difficulties remain. As we look towards a future shaped by VCTs, we must also be ready to navigate the complexities of this new paradigm.
Learn more about VCT challenges and opportunities here.
Practical Considerations for the Implementation of Virtual Clinical Trials
The implementation of virtual clinical trials requires meticulous planning and execution. This section offers insights for stakeholders considering the adoption of VCTs, with the goal of highlighting practical considerations at each step of the process—from participant recruitment to data gathering and interpretation.
- Identification of Clinically Meaningful Endpoints
- Determination of Optimal Sample Size
- Patient, Caregiver, and Site Training
- User Experience Optimization
- Device and Wearable Integration
- Notifications and Reminders
- Centralized Raters
- Real-Time Monitoring
1. Identification of Clinically Meaningful Endpoints
Before embarking on a VCT, it's crucial to define primary and secondary endpoints that reflect the direct impacts of the intervention on patient health. The quality of evidence and value of a study's outcomes should steer the direction of the trial, prompting researchers to measure the most relevant efficacy and safety signals of the intervention.
2. Determination of Optimal Sample Size
The sample size in a trial must be large enough to generate statistically significant results. Samples should also represent the diversity of the relevant population. Inclusion of participants of varying ages, ethnicities, and health statuses ensures the findings will be applicable to a broader population, improving the potential impact and generalizability of your research. A more restricted sample may be more appropriate when the indication or the targeted nature of the therapy warrants.
3. Patient, Caregiver, and Site Staff Training
VCT patients and caregivers are often key data collectors. It is therefore essential they receive thorough training on symptom reporting. Clear and concise instructions about the use of measurement tools can enhance data accuracy and reliability. In addition, well-informed patients are more likely to adhere to the trial protocol, improving the quality and integrity of the trial.
The site staff responsible for study coordination must also be properly trained to ensure they are intimately familiar with the trial objectives as well as the details of the protocol. In this way, they will be armed to support their patients and ensure smooth study progress. They should be able to answer patient questions, address concerns, and react to issues that arise in a timely manner.
4. User Experience Optimization
A positive user experience can significantly influence patient participation and adherence in a VCT. Trial design should strive for intuitive and user-friendly digital interactions, including app use, eConsent processes, and electronic patient-reported outcomes (ePROs). Ease of use and readability can minimize friction, making it more likely that patients will remain engaged throughout the trial.
5. Device and Wearable Integration
The use of clinical-grade devices or, in some cases, consumer wearables can contribute to the capture of reliable, real-time patient data with minimal effort from participants. Devices such as heart rate monitors, glucometers, or sleep trackers can provide a wealth of valuable information. Of course, the choice of devices should be carefully considered based on the trial's specific requirements.
6. Notifications and Reminders
Integration of digital notifications and reminders can improve patient engagement and ensure adherence to study protocols. Regular prompts can help patients remember to take medication, attend virtual visits, or report symptoms, contributing to better trial compliance and more accurate results.
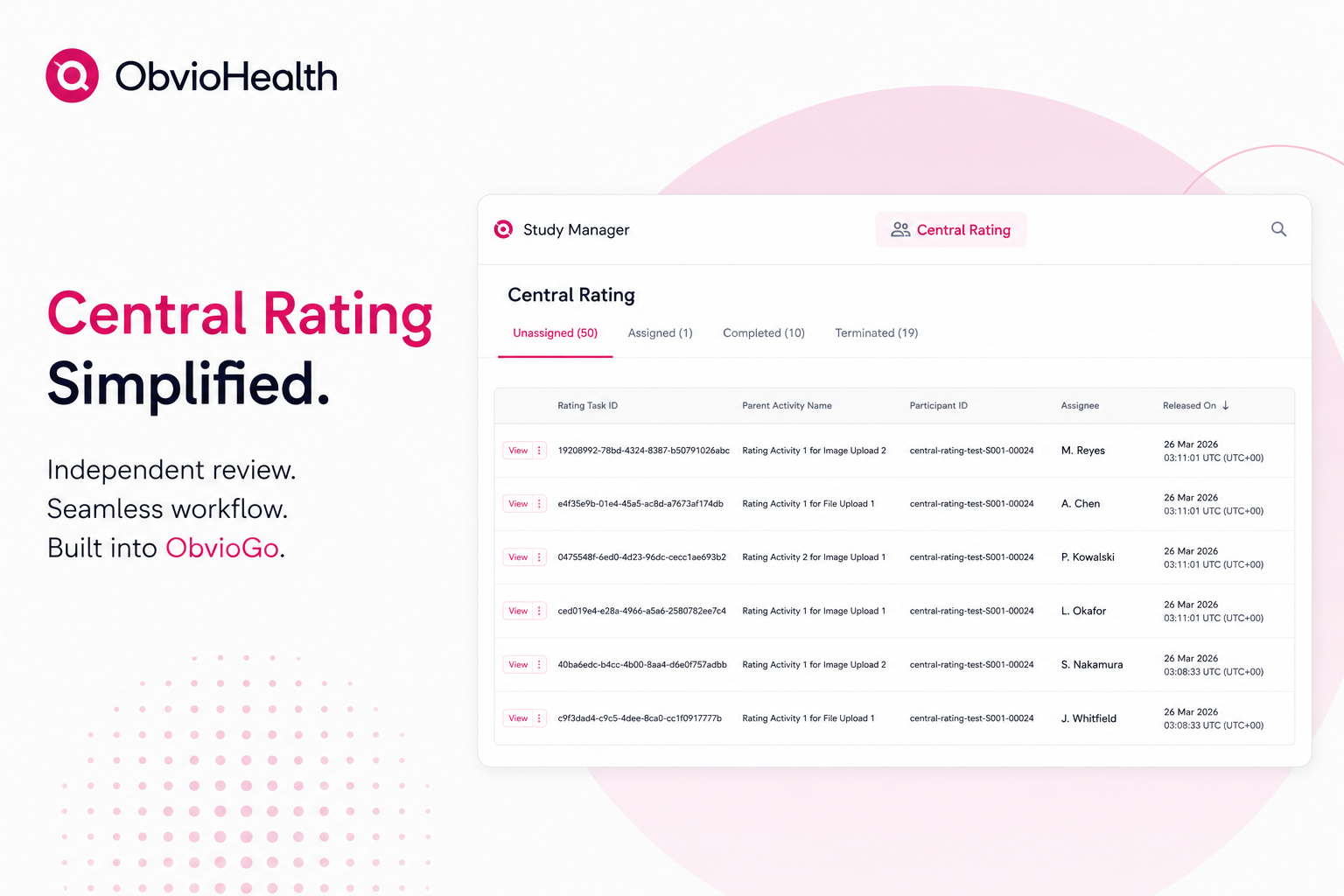
7. Centralized Raters
Centralized raters assess patient data remotely from a single location. Use of such raters can enhance the consistency and reliability of assessments in VCTs. By eliminating potential individual or institutional biases, centralized raters can help maintain the quality and integrity of the trial.
8. Real-Time Monitoring
Real-time monitoring in VCTs enables the prompt detection of issues, ensuring patient safety and maintaining study integrity. This includes monitoring patient compliance, adverse events, data quality, and more. By addressing concerns quickly, researchers can ensure the validity of the trial and protect participant well-being.
The above considerations should help sponsors initiate careful planning and thoughtful implementation that will lead to the successful outcomes of their virtual clinical trials. However, it would be naïve to imagine that the VCT universe will remain static. Future developments in the digitization and decentralization of clinical trials may have further implications for the research processes that support therapeutic innovation.

Looking Ahead: The Future of Virtual Clinical Trials
This section will discuss anticipated advancements in VCTs, including the potential use of artificial intelligence (AI), machine learning, digital health technologies (DHTs), and the broader implications of these innovations on the clinical research landscape.
The Power of AI, Machine Learning, and Digital Health Technologies
The intersection of healthcare and technology is catalyzing innovative solutions to long-standing challenges in clinical trials. AI and machine learning are already used to facilitate trial execution, and they can be expected to play a pivotal role in the future of VCTs, offering profound advancements in data collection and analysis. According to a Deloitte report, these cutting-edge technologies can enhance patient recruitment, optimize trial design, and help to predict trial outcomes.18 Furthermore, AI can enhance patient monitoring, allowing for real-time adjustments to treatment plans based on collected data.
Digital health technologies (DHTs) also hold immense potential in reshaping VCTs. DHTs—including sensors, wearables, and implantable devices, as well as more sophisticated mobile health applications—can collect valuable patient data in real time, improving the signal-to-noise ratio in studies to provide less biased, more precise data.1 Combining this with behavioral data from patient-reported outcomes will enable clinicians to form a more holistic view of the patient experience.
In a broader context, the advent of new digital technologies leveraged through VCTs will result in studies that can more easily adapt to specific patient diagnoses and symptoms, bringing the industry a step closer to the promise of personalized medicine.19
Conclusion: Countless Opportunities and Constructive Collaboration
As with most industries effected by rapid technological innovation, cross-industry consortia and partnerships will be critical to realizing the full potential of VCTs. There will be a need to align on data exchange standards, harmonization of methods, and, importantly, the development and validation of new promising instruments that can more precisely and effectively measure outcomes.
The Clinical Trials Transformation Initiative (CTTI) emphasizes the need for cross-industry partnerships to identify and address challenges and optimize processes in VCTs.20 By pooling resources, knowledge, and expertise, we can continue to drive forward the evolution of clinical trials, making them more efficient, accessible, and patient-friendly.
The future of VCTs is a collective effort—one where patients, researchers, technology providers, and regulators come together to harness the power of science and technology to help bring life-improving therapies to market more quickly and efficiently. We call on stakeholders across the health research ecosystem to embrace the opportunities offered by VCTs. By adopting these innovative trial methods and sharing best practices, we can usher in a new era of health innovation.
References
1. De Brouwer W, Patel CJ, Manrai AK, Rodriguez-Chavez IR, Shah NR. Empowering clinical research in a decentralized world. npj Digit Med. 2021;4(102). doi: 10.1038/s41746-021-00473-w
2. Miseta E. What Exactly Is A Virtual Clinical Trial? Clinical Leader. November 21, 2019. Accessed June 29, 2023. https://www.clinicalleader.com/doc/what-exactly-is-a-virtual-clinical-trial-0001
3. Chiang A, Herbst RS. How Telemedicine Can Transform Clinical Research and Practice. The ASCO Post. December 25, 2022. Accessed June 29, 2023. https://ascopost.com/issues/december-25-2022/how-telemedicine-can-transform-clinical-research-and-practice/
4. Ilancheran M. Use Of Wearable And Sensor Applications In Clinical Trials Is Booming. Clinical Leader. October 26, 2021. Accessed June 29, 2023. https://www.clinicalleader.com/doc/use-of-wearable-and-sensor-applications-in-clinical-trials-is-booming-0001
5. Ellis LD. Exploring the Promise of Wearable Devices to Further Medical Research. Trends in Medicine. May 19, 2023. Accessed June 30, 2023. https://postgraduateeducation.hms.harvard.edu/trends-medicine/exploring-promise-wearable-devices-further-medical-research
6. Adesoye T, Katz MHG, Offodile AC. Meeting Trial Participants Where They Are: Decentralized Clinical Trials as a Patient-Centered Paradigm for Enhancing Accrual and Diversity in Surgical and Multidisciplinary Trials in Oncology. JCO Oncol Pract. 2023;19(6):317-321. doi: 10.1200/op.22.00702
7. FDA. FDA Takes Additional Steps to Advance Decentralized Clinical Trials. 2023. Accessed June 27, 2023. https://www.fda.gov/news-events/press-announcements/fda-takes-additional-steps-advance-decentralized-clinical-trials
8. Forrest F. Virtual clinical trials: How digital tech has improved clinical trial accrual rates. Clinical Trials Arena. May 18, 2023. Accessed June 29, 2023. https://www.clinicaltrialsarena.com/sponsored/virtual-clinical-trials-how-digital-tech-has-improved-clinical-trial-accrual-rates/
9. Jiang S, Hong YA. Clinical trial participation in America: The roles of eHealth engagement and patient–provider communication. Digit Health. 2021;7. doi: 10.1177/20552076211067658
10. Fantana AL, Cella GM, Benson CT, Kvedar JC. The Future of Drug Trials Is Better Data and Continuous Monitoring. Harvard Business Review. May 2, 2019. Accessed June 28, 2023. https://hbr.org/2019/05/the-future-of-drug-trials-is-better-data-and-continuous-monitoring
11. DiMasi JA, Smith Z, Oakley-Girvan I, et al. Assessing the Financial Value of Decentralized Clinical Trials. Ther Innov Regul Sci. 2023;57:209-219. doi: 10.1007/s43441-022-00454-5
12. Costello M. Five Tips to Help Research Sites Implement Decentralized Clinical Trials. Applied Clinical Trials. 2020;29(10). https://www.appliedclinicaltrialsonline.com/view/five-tips-to-help-research-sites-implement-decentralized-clinical-trials
13. Benefits and Challenges of Virtual Clinical Trials. Clinical Research Society. July 17, 2019. Accessed June 28, 2023. https://www.clinicalresearchsociety.org/benefits-challenges-virtual-clinical-trials/
14. Vayena E, Blasimme A, Sugarman J. Decentralised clinical trials: ethical opportunities and challenges. Lancet Digit Health. 2023;5(6). doi: 10.1016/s2589-7500(23)00052-3
15. Telemedicine Can’t Get Safer Without Bridging the “Digital Divide.” April 1, 2022. Accessed June 30, 2023. https://www.ihi.org/communities/blogs/telemedicine-can-t-get-safer-without-bridging-the-digital-divide
16. Office of Research on Women’s Health. Review of the Literature: Primary Barriers and Facilitators to Participation in Clinical Research. National Institutes of Health. 2021. Accessed June 21, 2023. https://orwh.od.nih.gov/sites/orwh/files/docs/orwh_outreach_toolkit_litreview.pdf
17. FDA. Decentralized Clinical Trials for Drugs, Biological Products, and Devices: Guidance for Industry, Investigators, and Other Stakeholders. U.S. Department of Health and Human Services. 2023. Accessed June 23, 2023. https://www.fda.gov/media/167696/download
18. Morgan J. What is the future of virtual clinical trials? Deloitte. 2022. Accessed June 28, 2023. https://www2.deloitte.com/us/en/blog/health-care-blog/2022/what-is-the-future-of-virtual-clinical-trials.html
19. Agrawal G, Xue J, Moss R, Raschke R, Wurzer S. Stepping up the decentralization of clinical trials. McKinsey & Company. June 10, 2021. Accessed June 30, 2023. https://www.mckinsey.com/industries/life-sciences/our-insights/no-place-like-home-stepping-up-the-decentralization-of-clinical-trials
20. CTTI Vision for Clinical Trials in 2030. CTTI. 2020. Accessed June 12, 2023. https://ctti-clinicaltrials.org/who_we_are/transforming-trials-2030/